|
We can now look back at the electromagnetic spectrum above and realize that it is also ordered by energy gamma photons each carry more energy than ultraviolet photons and so on down the line. These particles, we call them photons, each carry a certain amount of energy, namely \ The energy of a photon, \(E\), is equal to Planck's constant, \(h\), (a number Max Planck found in his explanation of blackbody radiation, another of those minor issues physics was dealing with back then, and one we'll be seeing a lot of in this course) times the frequency, \(\nu\). The amplitude of the light is then proportional to. While the wave picture of light is good for many things, sometimes, like when trying to understand the photoelectric effect, it is more useful to think of light as being made of particles. According to Plancks equation, the energy of a photon is proportional to the frequency of the light. 
Indeed there is a characteristic threshold frequency for each metal below which there are no electrons ejected regardless of the intensity of the incident light (well, until you throw in so much energy you turn the metal into a plasma, but that is cheating, that is not the photoelectric effect).Īlbert Einstein won the Nobel Prize in Physics (1921) in part for his explanation of the photoelectric effect. If the wavelength is too large (the frequency too low) for the particular metal, no electrons will be ejected at all. Play around with the photoelectric effect simulation available at PhET you should find that while the number of electrons ejected depends on the intensity of the light, the energy of those electrons depends on the wavelength of the light. Important parameters, equations, and terms: Intensity, I: Transmittance, T Absorbance, A Beer-Lambert law Blank spectrum Cuvette Dark spectrum UV-Vis. One of those issues was the photoelectric effect, in which light striking a metal surface ejects electrons from the metal. While the above picture of light is all well and good, there were a few outstanding "issues" in physics at the end of the 19 th and beginning of the 20 th century. The basic principle is that each compound absorbs or transmits light over a certain range of wavelength. 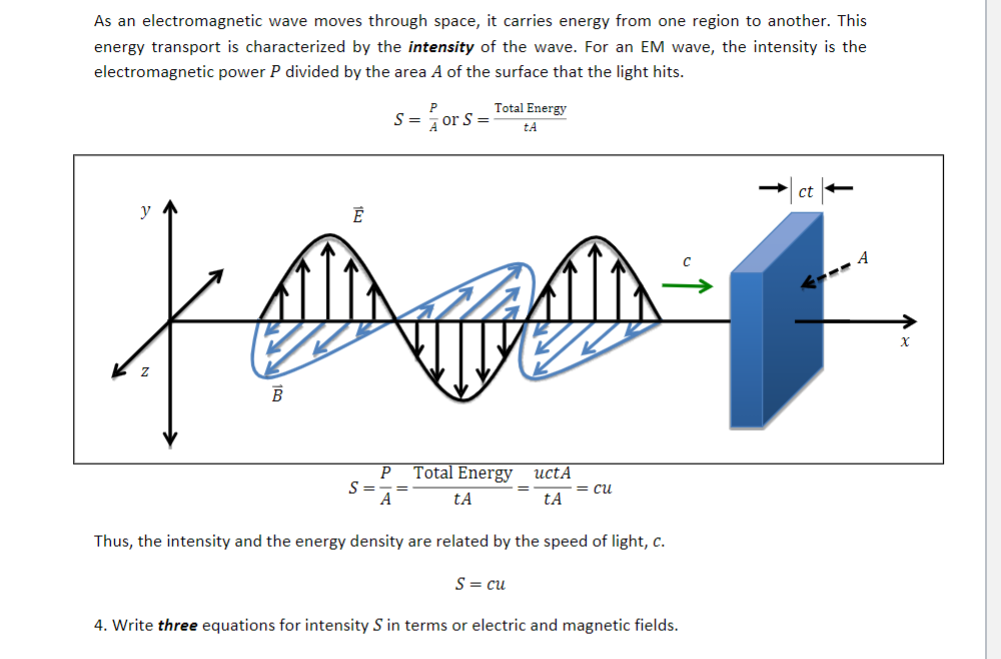 
Typical units of absorbance are called 'absorbance units,' which have the abbreviation AU. The property is measured using spectroscopy, particularly for quantitative analysis. It is also known as optical density, extinction, or decadic absorbance. Light is made of particles called photons Intensity is the quantity of energy the wave conveys per unit time across a surface of unit area, and it is also equivalent to the energy density multiplied by the wave speed. Spectrophotometry is a method to measure how much a chemical substance absorbs light by measuring the intensity of light as a beam of light passes through sample solution. Absorbance is a measure of the quantity of light absorbed by a sample.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
